Our Pipeline
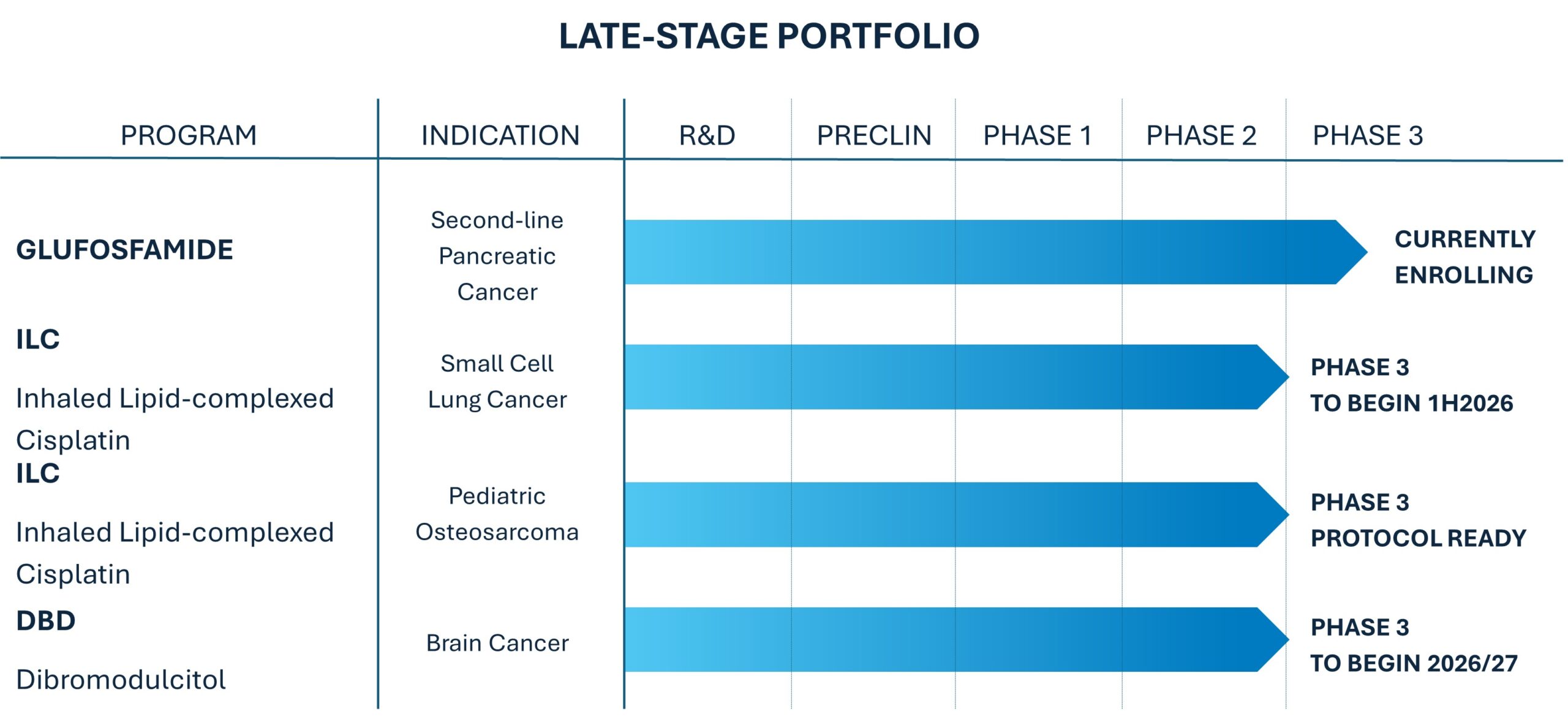

Glufosfamide

Several types of cancer, including pancreatic cancer, are more metabolically active than normal cells, requiring glucose as an energy source to fuel cell growth and division. These metabolically active cancers take up the glucose-conjugated glufosfamide, concentrating the active chemotherapeutic agent in cancer cells relative to normal tissue. In this way, glufosfamide targets cancer cells while reducing toxicity to normal cells.
Glufosfamide has been evaluated in more than 450 patients in multiple clinical studies and has shown clinical activity in a number of cancer types (including colon, breast, gall bladder, soft tissue). It was generally well tolerated with dose-limiting toxicity related to impairment of kidney function. Data from a previous Phase III trial from a prior sponsor indicated a 25% improvement in median survival in a very difficult to treat indication, second-line pancreatic cancer, although the study size and design of that trial were insufficient to prove statistical significance. Based on the valuable information learned from the prior study and with assistance from key clinical oncologist advisors, we designed a new pivotal Phase III study in second-line pancreatic cancer, which is currently underway and expected to be completed in 2027.
About Pancreatic Cancer
The American Cancer Society estimates more than 67,000 Americans are diagnosed with pancreatic cancer annually. It is a grim prognosis, with a five-year survival rate of only 5%, and accounted for an estimated 52,000 deaths in 2025 in the United States. The annual incidence in the European Union is approximately 80,000. Most patients present with metastatic disease and are not surgical candidates. Standard first-line therapy for most patients consists of gemcitabine chemotherapy alone or in combination with other agents. Unfortunately, many patients experience little or no benefit, and among responders, nearly all experience progressive disease in a few months. Currently, there are several options for second-line treatment, but there is no one standard of care for second-line treatment, and response rates and survival rates are minimal. Thus, there is an obvious clinical need for new drugs to treat patients with pancreatic cancer.

ILC
ILC technology is designed to offer three unique advantages in the treatment of lung disease over other existing inhalation therapies.
- The ability to attain a prolonged therapeutic effect of a drug in the lung by sustained release
- The ability to target and enhance the uptake of the drug into the site of diseased cells
- The ability to reduce systemic toxicities
ILC administered by inhalation may be effective for the treatment of certain lung cancers that can be reached by an inhaled chemotherapeutic agent. We believe that the unique characteristics of ILC could provide benefit to patients with non-metastatic SCLC in conjunction (as maintenance therapy) with first-line standard of care systemic chemotherapy to delay or prevent recurrence. Osteosarcoma patients with local (bone) disease may similarly benefit, as well as patients with pulmonary metastases at diagnosis, as palliative therapy for pulmonary metastases not amenable to complete resection surgically, or in combination with metastectomy to prevent or delay subsequent recurrences. Targeting of the lung with prolonged higher levels of cisplatin than can be achieved systemically could overcome relative cisplatin resistance and offer patients a prolonged disease-free interval or long-term survival.
ILC had been evaluated in 59 patients in three earlier clinical studies conducted by a prior sponsor with evidence of activity in several cancer types including lung and osteosarcoma, and in a 28-patient Phase II study of osteosarcoma that we conducted. In the four clinical studies, ILC was well tolerated in patients with no dose-limiting toxicity realized. In conjunction with an Asian partner, we anticipate conducting a Phase III study of ILC for patients with SCLC. Ultimately we hope to evaluate and develop ILC for other indications, including pediatric osteosarcoma, NSCLC and peritoneal metastatic cancers.
About Small Cell Lung Cancer
In the U.S. lung cancer is the second most common cancer type with 226,000 estimated new cases and the leading cause of cancer mortality with an estimated 125,000 deaths in 2025 (American Cancer Society). The two main types of lung cancer are non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). SCLC is far less common, comprising 10-15% of new lung cancer diagnoses. SCLC is typically treated with chemotherapy and most patients initially respond well to the treatment. Unfortunately, most patients will experience a recurrence or disease progression in a few month or more. Overall, five-year survival for SCLC patients is a dismal 5%-10%.
About Pediatric Osteosarcoma
In the U.S., osteosarcoma accounts for 5-10% of pediatric cancer each year - approximately 450 new cases among adolescents and young adults. The primary tumor site is typically in a leg bone, and usually not metastatic at time of diagnosis. Before the introduction of combination chemotherapy thirty years ago, amputation was the main treatment option and long term survival very low. Today, less radical surgery combined with a chemotherapy cocktail of methotrexate, doxorubicin, and cisplatin, results in an initial complete response in nearly all patients. Unfortunately, approximately 30-35% of patients will fail first-line therapy and will experience a recurrence several weeks to several years later. Most often, the site of recurrence is in the lungs and only in the lungs, with five-year survival less than 25%. By targeting the treatment and prevention of such pulmonary recurrences, ILC is potentially the first lifesaving breakthrough in the treatment of osteosarcoma in more than a generation.

DBD

DBD has been evaluated in more than 5,000 patients for diseases including brain cancer, breast cancer, cervical cancer, leukemia, and in cancers which metastasize to the brain. The primary findings included:
- Oral administration showed activity at a wide range of doses
- DBD crosses the blood-brain barrier
- DBD and be used in combination with radiation therapy
- DBD can be used in combination with certain other chemotherapy agents such as cisplatin
From a safety perspective, the major toxic effect of DBD is hematologic. It is dose related, and reversible. Other side effects reported included but not limited to nausea, vomiting, and hair loss.
The development plan for DBD requires completion of non-clinical and clinical bridging studies to demonstrate the safety equivalence of the new polymorph to original DBD polymorph. Upon completion of bridging studies, provided the data from such studies warrants, we will commence a pivotal Phase III registrational study.
About Brain Cancer
The American Cancer Society estimated that there will be more than 24,000 Americans diagnosed with brain cancer and cancer of the nervous system in 2025, and more than 18,000 related deaths. Long term survival is dependent on the sub-type of brain cancer and patient age, but for the glioblastoma multiforme sub-type, five year survival is only 5%. An estimated 290,000 persons are diagnosed with brain cancer and cancer of the nervous system each year on a worldwide basis (including the United States).